How long does it take uranium-235 to decay into lead 207?
How long does it take uranium-235 to decay into lead 207?
704 million years
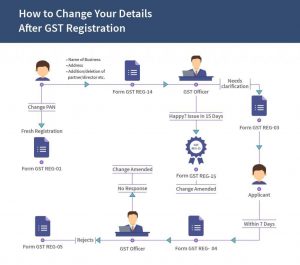
RADIOMETRIC TIME SCALE
| Parent Isotope | Stable Daughter Product | Currently Accepted Half-Life Values |
|---|---|---|
| Uranium-235 | Lead-207 | 704 million years |
| Thorium-232 | Lead-208 | 14.0 billion years |
| Rubidium-87 | Strontium-87 | 48.8 billion years |
| Potassium-40 | Argon-40 | 1.25 billion years |
How long does it take uranium-235 to decay?
700 million years
Uranium-235 has a half-life of just over 700 million years. Uranium-234 has the shortest half-life of them all at 245,500 years, but it occurs only indirectly from the decay of U-238. In comparison, the most radioactive element is polonium.
What does uranium-235 turn into when it decays?
Decay of uranium-235 into thorium-231 and an alpha particle. Larger, more massive nuclei like uranium-235 become more stable by emitting an alpha particle, which is a helium nucleus composed of two protons and two neutrons. This process is known as alpha decay.
What is the half-life of uranium-235?
about 700 million years
The half-life of uranium-238 is about 4.5 billion years, uranium-235 about 700 million years, and uranium-234 about 25 thousand years.
Does uranium 235 decay into lead?
Decay Chain of Isotope U-235 Beginning with the naturally-occurring isotope U-235, this decay series includes the following elements: Actinium, astatine, bismuth, francium, lead, polonium, protactinium, radium, radon, thallium, and thorium.
Can you hold plutonium in your bare hands?
A: Plutonium is, in fact, a metal very like uranium. If you hold it [in] your hand (and I’ve held tons of it my hand, a pound or two at a time), it’s heavy, like lead. It’s toxic, like lead or arsenic, but not much more so.
Why is U-235 better than u-238?
U- 235 is a fissile isotope, meaning that it can split into smaller molecules when a lower-energy neutron is fired at it. U- 238 has an even mass, and odd nuclei are more fissile because the extra neutron adds energy – more than what is required to fission the resulting nucleus.
Why is U-238 more stable than U-235?
The difference between the three isotopes is the number of neutrons present in the nucleus. U-238 has 4 more neutrons than U-234 and three more neutrons than U-235. U-238 is more stable thus being more abundant naturally. U-235 is used as fuel in nuclear reactors and/or weapons.
Why uranium-235 is unstable?
Uranium-235 (U-235) is one of the isotopes that fissions easily. During fission, U-235 atoms absorb loose neutrons. This causes U-235 to become unstable and split into two light atoms called fission products.
https://www.youtube.com/watch?v=F8sAkMQmOlU